BioUnfold #19 — Imaging at Scale: Homogeneity and Ambiguity in Discovery
Imaging has remained central to biological discovery for centuries, not because it captures everything, but because it preserves structure. Long before molecular measurements, biology advanced by looking: by observing morphology, spatial organization, and context. Modern imaging technologies are vastly more sophisticated, yet they retain this core property. They offer a system-level view that integrates many biological processes at once, without requiring early commitment to a specific molecular story.
At the same time, imaging only becomes operational in modern discovery when this richness is deliberately constrained.
What “scale” really means for imaging
In practice, imaging does not scale along a single dimension. It scales across multiple axes: the number of perturbations tested, the number of cells per condition, the number of features extracted per cell, the number of experimental batches, and the number of biological contexts compared. Pressure along any one of these axes forces compromise along others. Increasing the number of perturbations limits how much biological variability can be tolerated. Increasing biological heterogeneity increases ambiguity and reduces statistical efficiency. Scaling feature dimensionality complicates interpretation and stability.
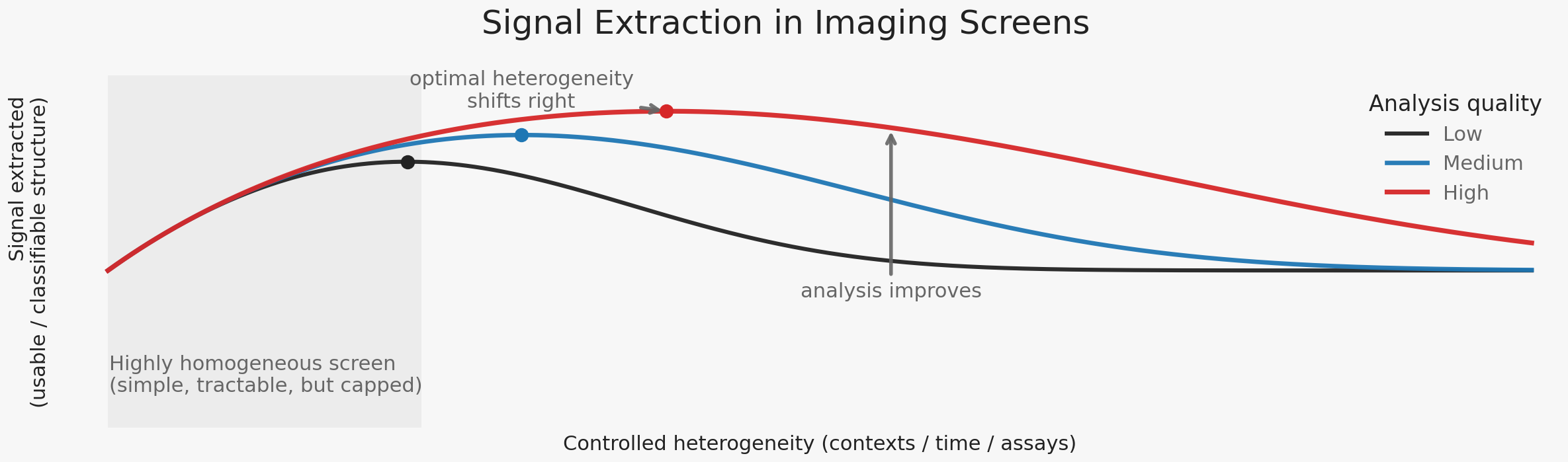
High-content imaging survives this pressure by enforcing homogeneity. Experimental systems are stabilized: single cell types, controlled density, fixed timepoints, standardized staining, synchronized states. These constraints collapse biological variability into a small number of dominant response modes. Metrics such as Z′ formalize this preference, rewarding experiments that maximize separation while minimizing variance. This is not an accident, nor a flaw. It is the mechanism by which imaging becomes reproducible, analyzable, and actionable at scale.
Why imaging works so well
The success of imaging screens follows directly from this design choice. Effect sizes are often large. Dose–response relationships are clean. Replicates cluster tightly. Simple models frequently suffice. These properties are sometimes attributed to the expressive power of imaging or to advances in analysis. More often, they reflect the fact that biology has been pushed into a small number of reproducible regimes.
This regime formation makes decisions possible. Compounds can be ranked. Mechanisms can be grouped. Programs can move forward. But homogeneity is not neutral. By stabilizing experiments, it freezes the space of distinctions the system is allowed to learn. When enforced too early or too rigidly, discovery becomes efficient but brittle—optimizing confidently within a narrow regime while remaining blind to alternative modes of response.
How heterogeneity was historically handled
Historically, heterogeneity was difficult to accommodate within imaging assays because it is expensive under throughput constraints. Allowing variability within a single experiment rapidly erodes effect sizes, collapses Z′, and increases the number of samples required to reach confidence. Discovery therefore scaled along a different axis: multiplying perturbations while holding biology fixed.
As a result, heterogeneity is introduced discretely across contexts — by testing hits in different cell lines, conditions, or assay variants — rather than being allowed to exist within primary screening experiments. This practice is most visible during hit confirmation and validation, where biological context is added stepwise and new distinctions are explicitly tested. Heterogeneity is deferred, not ignored.
This approach is not conceptually naïve. It reflects a pragmatic understanding that heterogeneity is only useful when it produces classifiable distinctions. Different biological contexts naturally terminate in labels: responder versus non-responder, selective versus non-selective, context-dependent versus general. Heterogeneity is discretized so that learning can proceed.
This remains the default today. What changes is not the logic, but the feasibility: improvements in analysis now make it possible to introduce heterogeneity earlier and more systematically, without abandoning decision discipline.
The asymptote, and why it matters now
In the limit, the ideal imaging experiment would observe multiple human tissues perturbed over time, preserving system balance while allowing statistical regularities to emerge before causal certainty. Such an experiment would be non-destructive, temporal, and context-rich. It would not immediately explain mechanisms, but it would reveal consistent patterns of response and failure across biological states.
For a long time, this ideal could only be approximated locally—through isolated assays, limited biological contexts, or carefully curated examples—because scaling heterogeneity systematically collapsed tractability. Today, that constraint is shifting. Advances in machine learning robustness, representation alignment, and assay standardization are changing the cost structure of ambiguity. Multiple complementary imaging experiments—across cell lines, timepoints, perturbations, and assay types—can increasingly be pursued deliberately and at scale. This does not remove ambiguity. It makes it manageable.
The consequence is not that discovery should abandon homogeneity, but that it can now afford to relax it strategically. Increased heterogeneity is justified only when it expands the space of classifiable hypotheses. Exploration can be broader, but it must still terminate in commitment.
Conclusion
Imaging does not advance discovery by preserving complexity indefinitely. It advances discovery by revealing new separations that can eventually be enforced. Homogeneity makes imaging work; heterogeneity keeps it honest. The discipline lies in knowing when to privilege one over the other.
The challenge is not to embrace heterogeneity for its own sake, but to introduce it deliberately—only where it sharpens hypotheses, and only until commitment becomes possible. In that balance, imaging remains not just a way of seeing biology, but a way of learning from it.