BioUnfold #26 — Two Engines, One Drug
Selling unfinished drugs to finance discovery
Modern drug discovery is organized around a paradox. The organizations that discover new therapeutics are rarely the ones that bring them to patients.
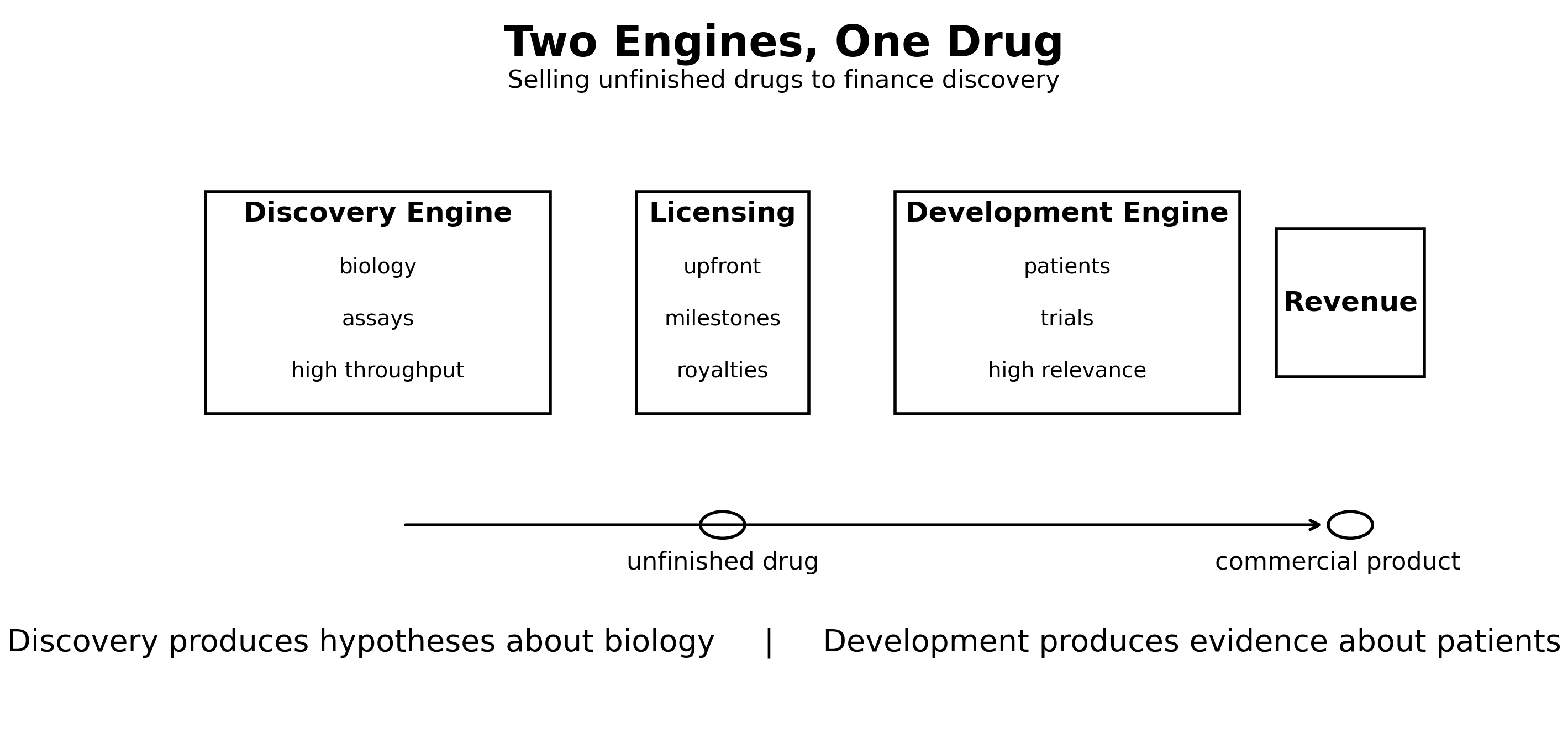
Biotech companies generate molecules. Pharmaceutical companies test them in clinical trials and commercialize them. Between those two steps lies licensing: the mechanism that connects discovery systems to development systems.
Licensing exists because no single organization is optimized for both discovery and development at the same time. Discovery and development operate at different speeds, require different capabilities, and tolerate different kinds of uncertainty. Licensing allows these two execution engines to cooperate on the same product while remaining specialized for different problems.
Drugs Are Sold Before Their Value Is Known
A therapeutic molecule is usually licensed long before its final value can be measured. At the time of a deal, the drug may not yet have entered late-stage clinical trials and may not even have been tested in humans. From preclinical discovery to approval, only about 5–10% of programs ultimately succeed.
This creates a valuation problem.
A successful drug may eventually generate billions in revenue. A failed drug generates none. Even strong preclinical evidence cannot resolve the gap between biological signal and patient outcome. The molecule exists in a state that is scientifically credible but economically indeterminate.
Licensing makes that uncertainty tradable.
Instead of assigning a single price to an unfinished product, the industry distributes value across time through upfront payments, milestones, and royalties. Each component transfers a different type of risk between organizations operating on different time horizons.
Biotech companies do not monetize drugs. They monetize credible progress toward drugs.
Discovery and Development Run at Different Speeds
The separation between biotech and pharmaceutical companies reflects the structure of the problem itself.
Discovery organizations operate through rapid experimental cycles. Assays evolve, hypotheses change, and programs are redirected as evidence accumulates. Progress depends on iteration and directional learning.
Clinical development operates differently. Trials must remain standardized across hospitals, regulators, and large patient populations over many years. Decisions cannot pivot easily once execution begins. Reliability becomes as important as insight.
Discovery optimizes exploration. Development optimizes consistency.
Licensing allows exploration and validation to remain separate without disconnecting them.
Upfront Payments Price Credibility
Upfront payments compensate discovery organizations for generating a molecule convincing enough to justify clinical investment despite the low overall probability of success.
At this stage, uncertainty is dominated by translation risk: whether signals observed in discovery systems will remain meaningful in humans. Upfront payments convert early credibility into immediate capital and allow discovery organizations to continue building their pipeline rather than waiting years for downstream validation.
They signal alignment between partners, but they do not resolve uncertainty. Upfront payments price belief rather than outcomes. Risk is shifted forward in time, not removed.
Milestones Distribute Risk Across Development
Milestones manage the uncertainty that remains after a partnership begins.
Instead of committing the full value of a drug early, partners agree on payments tied to events such as entering clinical phases, demonstrating efficacy, or reaching approval. Roughly half of drugs entering Phase I advance to Phase II. Only about one-third of Phase II programs reach Phase III. Even Phase III success is not guaranteed.
Each phase transition increases both confidence and cost.
Milestones allow development organizations to commit capital progressively as evidence strengthens, while allowing discovery organizations to recover investment earlier than commercialization would permit.
Evidence accumulates continuously, while milestone structures divide progress into contractual thresholds. Interpretation therefore becomes part of the partnership itself. Milestones redistribute uncertainty across time, but they do not remove it.
Royalties Preserve Exposure to Success
Royalties link discovery organizations to the long-term performance of a therapeutic after approval. They preserve exposure to outcomes that cannot be predicted at the time of licensing.
Some therapies expand far beyond their original expectations. Pembrolizumab (Keytruda), initially developed for melanoma, later became a backbone treatment across multiple cancers. At the time of early partnerships, this scale of impact could not have been anticipated.
Other drugs validate clinically but still underperform commercially. PCSK9 inhibitors demonstrated strong efficacy yet adoption remained constrained by pricing and reimbursement dynamics. Clinical success does not guarantee commercial dominance.
Royalties preserve exposure to success, but they arrive late and depend on execution outside the discovery organization’s control.
Exposure remains. Control does not.
Two Organizations, One Product
Licensing reflects something fundamental about how modern therapeutics are built.
Discovery organizations specialize in understanding biology. They design assays, generate hypotheses, and explore mechanisms using high-throughput systems that produce directional but indirect evidence about patients.
Development organizations operate closer to patients. They coordinate hospitals, regulators, and manufacturing across many years of execution. Their data is lower throughput but far more directly relevant to therapeutic outcomes.
Only some capabilities, such as chemistry optimization and manufacturing preparation, naturally span both environments.
Discovery produces hypotheses about biology. Development produces evidence about patients.
Licensing connects systems optimized for exploration with systems optimized for validation. Modern drug discovery depends on both